Photoelectric EffectHard
Question
The orbital speed of the electron in the ground state of hydrogen is v. What will be its orbital speed when it is excited to the energy state − 3.4 eV ?
Options
A.2 v
B.v/2
C.v/4
D.v/8
Solution
E ∝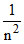
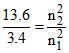 n2 = 2
n2 = 2
v ∝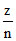 (z = 1) ⇒ v2 =
(z = 1) ⇒ v2 = 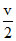 ....... (B)
....... (B)
v ∝
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
When X-rays tube is operated at V0 voltage, it produces Kα X-ray of wavelength λ. Now voltage is made then wav...What is the minimum wavelength of X-rays...The intensity of direct sunlight on a surface normal to the rays is I0. What is the intensity of direct sunlight on a su...This question has Statement - 1 and Statement - 2. Of the four choices given after the statements, choose the one that b...Which one of the following graphs in figure shows the variation of photoelectric current (I) with voltage (V) between th...