JEE Main | 2014Photoelectric EffectHard
Question
The radiation corresponding to 3 → 2 transition of hydrogen atoms falls on a metal surface to produce photoelectrons. These electrons are made to enter a magnetic field of 3 × 10-4 T. If the radius of the largest circular path followed by these electrons is 10.0 mm, the work function of the metal is close to
Options
A.1.8 eV
B.1.1 eV
C.0.8 eV
D.1.6 eV
Solution
r 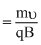
=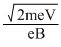
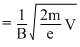
⇒ V =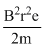 = 0.8 V
= 0.8 V
For transition between 3 to 2,
E = 13.6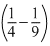
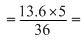 1.88 eV
1.88 eV
Work function = 1.88 eV - 0.8 eV
= 1.08 eV = 1.1 eV
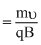
=
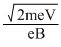
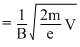
⇒ V =
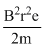 = 0.8 V
= 0.8 V For transition between 3 to 2,
E = 13.6
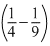
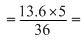 1.88 eV
1.88 eV Work function = 1.88 eV - 0.8 eV
= 1.08 eV = 1.1 eV
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
A given shaped glass tube having uniform cross-section is filled with water and is mounted on a rotatable shaft as shown...A body has 60kg weight. He wants to swim in a river with the help of a wooden log. If relative density of wood is 0.6 wh...When light of wavelength lesser than 6000 is incident on a metal, electrons are emitted. The approximate work-function o...The rain drops are in spherical shape due to...The wavelength of the first line in balmer series in the hydrogen spectrum is λ. What is the wavelength of the seco...