KTGHard
Question
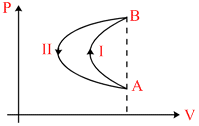
In a cyclic process, a gas is taken from state A to B via path-I as shown in the indicator diagram and taken back to state A from state B via path-II. In the complete cycle :

Options
A. work is done on the gas.
B.heat is given to the gas
C.no work is done by the gas.
D.nothing can be said about work as data is insufficient
Solution
For anticlockwise, work is negative i.e., work is done on the gas, hence option A and C are correct.

Create a free account to view solution
View Solution FreeMore KTG Questions
Two thermally insulated vessels 1 and 2 are filled with air at temperatures (T1, T2), volume (V1, V2) and pressure (P1, ...Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will...For an ideal gas, the heat capacity at constant pressure is larger than than that at constant volume because...A student performs an experiment to determine the Young′s modulus of a wire, exactly 2 m long, by Searle′s m...A one dimensional gas is a hypothetical gas with molecules that can move along only a single axis. The following table g...