KTGHard
Question
Two thermally insulated vessels 1 and 2 are filled with air at temperatures (T1, T2), volume (V1, V2) and pressure (P1, P2) respectively. If the valve joining two vessels is opened, the temperature inside the vessel at equilibrium will be
Options
A.T1 + T2
B.(T1 + T2)/2
C.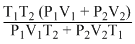
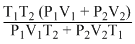
D.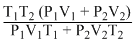
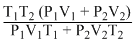
Solution
The number of moles of system remains same
According to Boyle′s law,
P1V1 + P2V2 = P(V1 + V2)
∴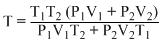
According to Boyle′s law,
P1V1 + P2V2 = P(V1 + V2)
∴
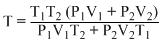
Create a free account to view solution
View Solution FreeMore KTG Questions
Root mean square velocity for a certain di-atomic gas at room temperature 27oC is found to be 1930 m/sec. The gas is -...Air is filled at 60o C in a vessel of open mouth. The vessel in heated to a temperature T so that 1/4th part of air esca...When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the ...Following is not a postulate of kinetic theory of gases :-...An ideal gas at 1 atmospheric pressure and at 273 K has 22.4 litre of volume. This is heated to 546 K and then by applyi...