KTGHard
Question
When a sample of a gas is taken from state i to state f along path ′iaf′, heat supplied to the gas is 50 cal and work done by the gas is 20 cal. If it is taken by path ′ibf′ , then heat supplied is 36 cal.
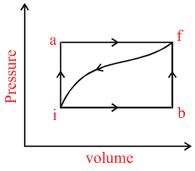

Options
A.Work done by the gas along path ibf is 6 cal
B.If work done on the gas is 13 cal for the return path ′ fi ′, then heat rejected by the gas along path ′ fi ′ is 43 cal.
C.If internal energy of the gas at state i is 10 cal, then internal energy at state ′f′ is 40 cal.
D.If internal energy at state ′b′ is 22 cal and at ′i′ is 10 cal then heat supplied to the gas along path ′ib′ is 18 cal.
Solution
Uf − Ui = dU = Q − W = 50 − 20 = 30 cal.
Qibf = 36 cal, dU = 30 cal.
Therefore Wibf = Qibf − dU = 6 cal.
For path ′fi′ dU = − 30 cal
W = − 13 cal.
Q = − 30 − 13 = − 43 cal
Negative Q means heat has been rejected by the gas.
Again as Uf − Ui = 30 cal and Ui = 10 cal
Then Uf = 40 cal.
Along path ′ibf′ W = 6 cal
again as Wbf = 0
therefore Wib = 6 cal.
Ub − Ui = 22 − 10 = 12 cal.
Hence Q = Wib + (Ub − Ui) = 18 cal.
Qibf = 36 cal, dU = 30 cal.
Therefore Wibf = Qibf − dU = 6 cal.
For path ′fi′ dU = − 30 cal
W = − 13 cal.
Q = − 30 − 13 = − 43 cal
Negative Q means heat has been rejected by the gas.
Again as Uf − Ui = 30 cal and Ui = 10 cal
Then Uf = 40 cal.
Along path ′ibf′ W = 6 cal
again as Wbf = 0
therefore Wib = 6 cal.
Ub − Ui = 22 − 10 = 12 cal.
Hence Q = Wib + (Ub − Ui) = 18 cal.
Create a free account to view solution
View Solution FreeMore KTG Questions
A fixed container is fitted with a piston which is attached to a spring of spring constant k. The other end of the sprin...A diatomic gas molecules has translational, rotational and vibrational degrees of freedom, then ratio is...At a given temperature, the pressure of an ideal gas of density ρ is proportional to -...A diatomic ideal gas undergoes a thermodynamic change according to the P−V diagram shown in the figure. The total ...What is critical temperature, If a and b are vander wall′s constant....