KTGHard
Question
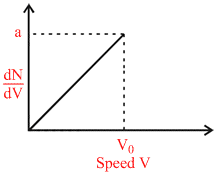
Graph shows a hypothetical speed distribution for a sample of N gas particle (for V > V0; 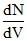 = 0)
= 0)

Options
A.The value of aV0 is 2N.
B.The ratio Vavg/V0 is equal to 2/3.
C.The ratio Vrms/V0 is equal to 1/√2.
D.Three fourth of the total particle has a speed between 0.5 V0 and V0.
Solution
Area under the curve is equal to number of molecules of the gas sample. Hence
N =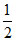 . a . V0 ⇒ aV0 = 2N
. a . V0 ⇒ aV0 = 2N
Vavg =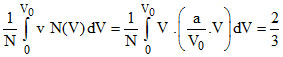 V0 ⇒
V0 ⇒ 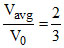
vrms2 =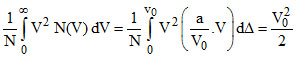 ⇒
⇒ 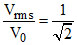
Area under the curve from 0.5 V0 to V0 is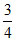 of total area.
of total area.
N =
Vavg =
vrms2 =
Area under the curve from 0.5 V0 to V0 is
Create a free account to view solution
View Solution FreeMore KTG Questions
In above question, if Ud = 88 J, heat absorbed for the path ′db′ is...Root mean square velocity for a certain di-atomic gas at room temperature 27oC is found to be 1930 m/sec. The gas is -...In a cyclic process, a gas is taken from state A to B via path-I as shown in the indicator diagram and taken back to sta...Let ᐃW1 and ᐃW2 be the work done by the systems 1 and 2 respectively in the previous question then :...The quantities which remains same for all ideal gases at the same temperature is/are ?...