KTGHard
Question
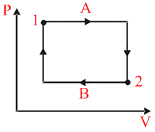
In given figure, let ᐃU1 and ᐃU2 be change in internal energy in process A and B respectively. ᐃQ and W be the net heat given and net work done by the system in the process A + B, then

Options
A.ᐃU1 + ᐃU2 = 0
B.ᐃU1 − ᐃU2 = 0
C.ᐃQ − W = 0
D.ᐃQ + W = 0
Solution
in cyclic process ᐃU1 + ᐃU2 = 0 ᐃUNet = 0 ᐃQ − ᐃW = 0
Create a free account to view solution
View Solution FreeMore KTG Questions
Gases obey vander -waal′s equation at :...When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the ...An ideal gas undergoes a thermodynamic cycle as shown in figure.:Which of the following graphs represents the same cycle...A fixed mass of gas undergoes the cycle of changes represented by PQRSP as shown in Figure. In some of the changes, work...Ideal gas is taken through process shown in figure:...