KTGHard
Question
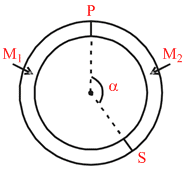
A ring shaped tube contains two ideal gases with equal masses and atomic mass numbers M1 = 32 and M2 = 28. The gases are separated by one fixed partition P and another movable conducting partition S which can move freely without friction inside the ring. The angle a as shown in the figure (in degrees) in equilibrium is:

Options
A.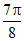
B.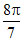
C.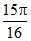
D.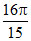
Solution
P1 = P2 [In equilibrium]
T1 = T2 ⇒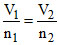 ⇒
⇒ 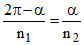
⇒ M1(2π − α) = M2 α
or a =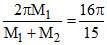
T1 = T2 ⇒
⇒ M1(2π − α) = M2 α
or a =
Create a free account to view solution
View Solution FreeMore KTG Questions
Three moles of an ideal monoatomic gas perform a cycle shown in figure. The gas temperatures in different states are T1 ...Gases obey vander -waal′s equation at :...A fixed container is fitted with a piston which is attached to a spring of spring constant k. The other end of the sprin...The ratio of the specific heats = γ in terms of degrees of freedom (n) is given by :...Gases O2 and H2 filled in a container the ratio at their r.m.s. velocity -...