PYQKTGHard
Question
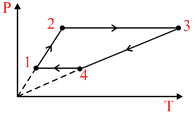
Three moles of an ideal monoatomic gas perform a cycle shown in figure. The gas temperatures in different states are T1 = 400 K, T2 = 800 K, T3 = 2400 K, and T4 = 1200 K. The work done by the gas during the cycle is :- 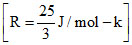

Options
A.5 kJ
B.10 kJ
C.15 kJ
D.20 kJ
Solution
(D)
Process1→2 and Process3→4 are isochoric process.
W12 = 0
W34 = 0
W23 = n R (T3 − T2)
= 3 R (2400 − 800) = 4800 R
W41 = nR (T1 − T4)
= 3 R (400 − 1200) = − 2400 R
W = (4800 − 2400) R = 2400 R
= 20 kJ
Process1→2 and Process3→4 are isochoric process.
W12 = 0
W34 = 0
W23 = n R (T3 − T2)
= 3 R (2400 − 800) = 4800 R
W41 = nR (T1 − T4)
= 3 R (400 − 1200) = − 2400 R
W = (4800 − 2400) R = 2400 R
= 20 kJ
Create a free account to view solution
View Solution FreeMore KTG Questions
An ideal gas undergoes a thermodynamic cycle as shown in figure.:Which of the following graphs represents the same cycle...Let ᐃW1 and ᐃW2 be the work done by the systems 1 and 2 respectively in the previous question then :...The temperature at which root mean square velocity of molecules of helium is equal to root mean square velocity of hydro...When an ideal gas undergoes an adiabatic change causing a temperature change ᐃT(i) there is no heat gained or lost...The root mean square speed of the molecules of a gas is :...