KTGHard
Question
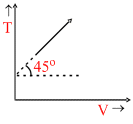
The given curve represents the variation of temperature as a function of volume for one mole of an ideal gas. Which of the following curves best represents the variation of pressure as a function of volume?

Options
A.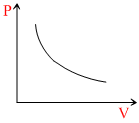

B.

C.

D.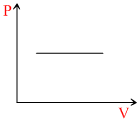

Solution
Here, T = V tan45o + T0 ⇒ T = V + T0
And P =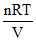
∴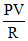 = V + T0 (Since, n = 1)
= V + T0 (Since, n = 1)
∴ (P − R)V = RT0
Therefore, Graph will be rectangular hyperbola.
And P =
∴
∴ (P − R)V = RT0
Therefore, Graph will be rectangular hyperbola.
Create a free account to view solution
View Solution FreeMore KTG Questions
The rms velocity of gas molecules of a given amount of a gas at 27o C and 1.0 × 105 Nm-2 pressure is 200 m sec-1. I...If the pressure of a gas is doubled with constant temperature, then the mean square velocity will become :-...If K is Boltzman′s constant, m the mass of one molecule and temperature of gas is T, then root mean square velocit...In a sample of an ideal gas the average momentum of a molecule depends on...Average kinetic energy of a gas molecule depends on-...