KTGHard
Question
The rms velocity of gas molecules of a given amount of a gas at 27o C and 1.0 × 105 Nm-2 pressure is 200 m sec-1. If temperature and pressure are respectively 127o C and 0.5 × 105 Nm2, the rms velocity will be :-
Options
A.400 / √3 ms-1
B.100 / √2 ms-1
C.100 √2 / 3ms-1
D.50 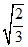 ms-1
ms-1
More KTG Questions
A thermally insulated chamber of volume 2V0 is divided by a frictionless piston of area S & mass m into two equal parts ...The average speed of nitrogen molecules in a gas is v. If the temperature is doubled and the N2 molecule dissociate into...The constant quantity for equal volume of two gases at same pressure and temperature is-...For one gram mole of a gas, the gas equation is PV = RT, here ′R′ is :-...The Energy of Monoatomic gas is :-...