KTGHard
Question
In given figure, a fixed mass of an ideal gas undergoes the change represented by XYZX below. Which one of the following sets could describe these of changes ?
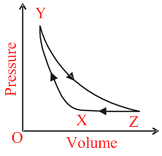

Options
A.XY → isothermal expansion
Y Z → adiabatic compression
Z X → compression at constant pressure
Y Z → adiabatic compression
Z X → compression at constant pressure
B.XY → adiabatic expansion
Y Z → isothermal compression
Z X → constant volume compression at
Y Z → isothermal compression
Z X → constant volume compression at
C.XY → isothermal compression
Y Z → adiabatic expansion
Z X → compression at constant pressure
Y Z → adiabatic expansion
Z X → compression at constant pressure
D.XY → adiabatic compression
Y Z → isothermal expansion
Z X → compression at constant pressure
Y Z → isothermal expansion
Z X → compression at constant pressure
Solution
XY Adiabatic compresion
YZ Isothermal Expansion
ZX Compression at constant pressure
YZ Isothermal Expansion
ZX Compression at constant pressure
Create a free account to view solution
View Solution FreeMore KTG Questions
In a sample of an ideal gas the average momentum of a molecule depends on......The specific heat of an ideal gas depends on temperature is-...In given figure, let ᐃU1 and ᐃU2 be change in internal energy in process A and B respectively. ᐃQ and ...Refer to fig. Let ᐃU1 and ᐃU2 be the changes in internal energy of the system in the processes A and B then...