KTGHard
Question
Refer to fig. Let ᐃU1 and ᐃU2 be the changes in internal energy of the system in the processes A and B then
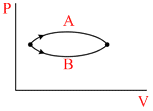

Options
A.ᐃU1 > ᐃU2
B.ᐃU1 = ᐃU2
C.ᐃU1 < ᐃU2
D.ᐃU1 ≠ ᐃU2
Solution
As ᐃU is a state function i.e., it depends initial and final position
∴ ᐃU, = ᐃU2 .
∴ ᐃU, = ᐃU2 .
Create a free account to view solution
View Solution FreeMore KTG Questions
If an ideal gas compressed during isothermal process then :-...If the root mean square speed of hydrogen molecules is equal to root mean square speed of oxygen molecules at 47oC, the ...Container x has volume double that of container y and both are connected by a thin tube. Both contains same ideal gas. T...Four curves A, B, C and D are drawn in the Fig. for a given amount of gas. The curves which represent adiabatic and isot...A vessel has 6g of oxygen of pressure P and temperature 400 K, a small hole is made in it so that oxygen leaks out. How ...