KTGHard
Question
Four curves A, B, C and D are drawn in the Fig. for a given amount of gas. The curves which represent adiabatic and isothermal changes are
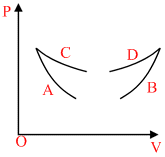

Options
A.C and D respectively
B.D and C respectively
C.A and B respectively
D.B and A respectively
Solution
Isothermal P ∝ 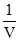
Adiabatic P ∝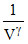
Also, slope of adiabatic is more as compare to isothermal
∴ option (C) is correct.
Adiabatic P ∝
Also, slope of adiabatic is more as compare to isothermal
∴ option (C) is correct.
Create a free account to view solution
View Solution FreeMore KTG Questions
On giving equal amount of heat to equal amount of a monoatomic and a diatomic gas the rise in temperature (ᐃT) is ...Refer to fig. Let ᐃU1 and ᐃU2 be the changes in internal energy of the system in the processes A and B then...The expansion of an ideal gas of mass m in volume at a constant pressure P is given by the straight line B. Then the exp...Ratio of molecular specific heats of a di- atomic gas will be :-...A gas is contained in a metallic cylinder fitted with a piston. The gas is suddenly compressed by pushing piston downwar...