KTGHard
Question
Let P1 and P2 be the final pressure of the samples 1 and 2 respectively in the previous question then :
Options
A.P1 < P2
B.P1 = P2
C.P1 > P2
D.The relation between P1 and P2 cannot be deduced.
Solution
For isothermal
PV = C . or
PV = C . or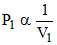 ...(i)
...(i)
For adiabatic
PVγ = C,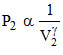 ...(ii)
...(ii)
from (i) and (ii)
P1 > P2
PV = C . or
PV = C . or
For adiabatic
PVγ = C,
from (i) and (ii)
P1 > P2
Create a free account to view solution
View Solution FreeMore KTG Questions
Three closed vessels A, B, and C are at the same temperature T and contain gases which obey the Maxwell distribution of ...At what temperature will the rms velocity of O2 molecules be equal to that of H2 at 200K...The root mean square speed of hydrogen molecules of an ideal hydrogen gas kept in a gas chamber at 0oC is 3180 m/s. The ...A gas behaves more closely like an ideal gas at...Three perfect gases at absolute temperatures T1, T2 and T3 are mixed. The masses of molecules are m1, m2 and m3 and the ...