KTGHard
Question
Three closed vessels A, B, and C are at the same temperature T and contain gases which obey the Maxwell distribution of speed . Vessel A contains only O2, B only N2 and C a mixture of equal quantities of O2 and N2. If the average speed of O2 molecules in vessel A is V1, that of the N2 molecules in vessel B is V2 ,the average speed of the O2 molecules in vessel C will be :
Options
A.(V1 + V2)/2
B.V1
C.(V1V2)1/2
D.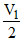
Solution
Vav = 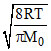 , VAV α √T
, VAV α √T
For same temp in vessel A, B and C, Average speed of O2 molecule is same in vessel A and C and is equal to V1.
For same temp in vessel A, B and C, Average speed of O2 molecule is same in vessel A and C and is equal to V1.
Create a free account to view solution
View Solution FreeMore KTG Questions
On giving equal amount of heat to equal amount of a monoatomic and a diatomic gas the rise in temperature (ᐃT) is ...The temperature at which the r.m.s velocity of oxygen molecules equal that of nitrogen molecules at 100oC is nearly:...Boiling water is changing into steam. Under this condition, the specific heat of water is...At a given temperature, the pressure of an ideal gas of density ρ is proportional to -...The mean kinetic energy of a molecule of an ideal gas is :...