KTGHard
Question
An ideal gas of one mole is kept in a rigid container of negligible heat capacity. If 25 J of heat is supplied the gas temperature raises by 20 C. Then the gas may be
Options
A.helium
B.argon
C.oxygen
D.carbon dioxide
Solution
ᐃQ = ᐃU + ᐃW ⇒ 25 = 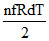 + 0
+ 0
25 =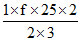
f = 3 (monoatomic)
25 =
f = 3 (monoatomic)
Create a free account to view solution
View Solution FreeMore KTG Questions
A balloon containing an ideal gas has a volume of 10 liter and temperature of 17oC. If it is heated slowly to 75oC, the ...Which of the following formula is wrong -...Critical temperature is that temperature...In a mixture of nitrogen and helium kept at room tempertaure. As compared to a helium molecule nitrogen molecule hits th...For a real gas, the following is true -...