KTGHard
Question
A balloon containing an ideal gas has a volume of 10 liter and temperature of 17oC. If it is heated slowly to 75oC, the work done by the gas inside the balloon is (neglect elasticity of the balloon and take atmospheric pressure as 105 Pa)
Options
A.100 J
B.200 J
C.250 J
D.data insufficient
Solution
Since elasticity of balloon is negligible ∴ pressure inside balloon pressure outside balloon = Patm.
∴ W = Patm ᐃV
Vin = 10 litre.
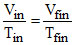 ⇒ Vfinal =
⇒ Vfinal = 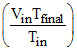 ltre. ⇒ W = Patm Vin
ltre. ⇒ W = Patm Vin 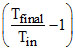 ⇒ 105 × 10-2
⇒ 105 × 10-2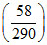 = 200 J
= 200 J
∴ W = Patm ᐃV
Vin = 10 litre.
Create a free account to view solution
View Solution FreeMore KTG Questions
Find the coorect relation in given P-V diagram :...Gases O2 and H2 filled in a container the ratio at their r.m.s. velocity -...The specific heat of an ideal gas depends on temperature is-...The temperature of ideal gas is kept same during the expansion. Gas is doing work. The internal energy of the gas during...Ratio of molecular specific heats of a di- atomic gas will be :-...