KTGHard
Question
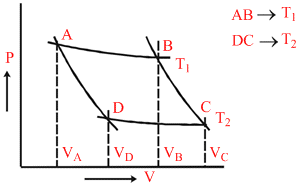
In the following P−V diagram of an ideal gas, AB and CD are isothermal where as BC and DA are adiabatic process. The value of VB/VC is

Options
A.= VA /VD
B.< VA /VD
C.> VA /VD
D.cannot say
Solution
AB → isothermal lerkih;
PA VA = PB VB ...(i)
BC → Adiabatic
PB VBγ = PC VCγ ...(ii)
CD → Isothermal
PCVC = PDVD ...(iii)
DA → Adiabatic
PDVDγ = PA VAγ ...(iv)
From (i), (ii), (iii) and (iv)
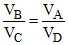
PA VA = PB VB ...(i)
BC → Adiabatic
PB VBγ = PC VCγ ...(ii)
CD → Isothermal
PCVC = PDVD ...(iii)
DA → Adiabatic
PDVDγ = PA VAγ ...(iv)
From (i), (ii), (iii) and (iv)
Create a free account to view solution
View Solution FreeMore KTG Questions
The kinetic energy associated with per degree of freedom of a molecule is-...In a mixture of nitrogen and helium kept at room tempertaure. As compared to a helium molecule nitrogen molecule hits th...What is critical temperature, If a and b are vander wall′s constant....N(< 100) molecules of a gas have velocities 1, 2, 3........ N km/s respectively. Then...At absolute zero temperature :-...