KTGHard
Question
When an ideal diatomic gas is heated at constant pressure , the fraction of the heat energy supplied which increases the internal energy of the gas is .
Options
A.2/5
B.3/5
C.3/7
D.5/7
Solution
dQ = dW + dU
dQ = PdV + dU
dQ = nRdT + dU
dQ =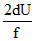 + dU
+ dU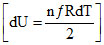
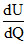 =
= 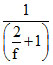
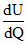 =
= 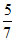
dQ = PdV + dU
dQ = nRdT + dU
dQ =
Create a free account to view solution
View Solution FreeMore KTG Questions
The adiabatic bulk modulus of hydrogen gas (γ = 1.4) at NTP is :...The total kinetic energy of 1 mole of N2 at 27o C will be approximately :-...In the following P−V diagram of an ideal gas, AB and CD are isothermal where as BC and DA are adiabatic process. T...A vessel has 6g of oxygen of pressure P and temperature 400 K, a small hole is made in it so that oxygen leaks out. How ...N(< 100) molecules of a gas have velocities 1, 2, 3........ N km/s respectively. Then...