KTGHard
Question
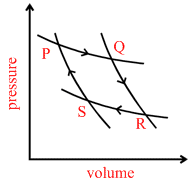
A fixed mass of gas undergoes the cycle of changes represented by PQRSP as shown in Figure. In some of the changes, work is done on the gas and in others, work is done by the gas. In which pair of the changes work is done on the gas?

Options
A.PQ and RS
B.PQ and QR
C.OR and RS
D.RS and SP.
Solution
work done on the gas = negative work
W = PdV when V → decreases
then W = − ve
hence option D is correct.
W = PdV when V → decreases
then W = − ve
hence option D is correct.
Create a free account to view solution
View Solution FreeMore KTG Questions
If a container is filled with the mixture of H2 and O2 then :-...One mole of a gas is subjected to two process AB and BC, one after the other as shown in the figure. BC is represented b...At temperature T, N molecules of gas A each having mass m and at the same temperature 2 N molecules of gas B each having...The temperature at which root mean square velocity of molecules of helium is equal to root mean square velocity of hydro...In isothermal process if heat is released from an ideal gas then,...