KTGHard
Question
A system undergoes a cyclic process in which it absorbs Q1 heat and gives out Q2 heat. The efficiency of the process is h and work done is W. Select correct statement:
Options
A.W = Q1 − Q2
B.η = 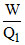
C.η = 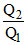
D.η = 1 − 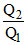
More KTG Questions
Find the amount of work done to increase the temperature of one mole of ideal gas by 30oC. if it is expanding under the ...If a container is filled with the mixture of H2 and O2 then :-...A balloon containing an ideal gas has a volume of 10 liter and temperature of 17oC. If it is heated slowly to 75oC, the ...A vessel has 6g of oxygen of pressure P and temperature 400 K, a small hole is made in it so that oxygen leaks out. How ...A piece of iron is heated in a flame. It first becomes dull red then becomes reddish yellow and finally turns to white h...