KTGHard
Question
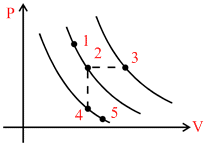
A certain gas is taken to the five states represented by dots in the graph. The plotted lines are isotherms. Order of the most probable speed vp of the molecules at these five states is :

Options
A.VP at 3 > VP at 1 = VP at 2 > VP at 4 = VP at 5
B.VP at 1 > VP at 2 = VP at 3 > VP at 4 > VP at 5
C.VP at 3 > VP at 2 = VP at 4 > VP at 1 > VP at 5
D.Insufficient information to predict the result.
Solution
Stages 1 and 2 are at same temperature also stages 4 and 5 are at same temperature.
As, VP is more at higher temperature and same at all stages at equal temperature.
∴ VP3 > VP1 = VP2 > VP4 = VP5 (As T3 > T2 = T1 > T4 = T5)
As, VP is more at higher temperature and same at all stages at equal temperature.
∴ VP3 > VP1 = VP2 > VP4 = VP5 (As T3 > T2 = T1 > T4 = T5)
Create a free account to view solution
View Solution FreeMore KTG Questions
If a container is filled with the mixture of H2 and O2 then :-...Suppose a container is evacuated to leave just one molecule of a gas in it. Let νmp and νav represent the most...One mole of a gas is subjected to two process AB and BC, one after the other as shown in the figure. BC is represented b...The pressure exerted by a gas in P0. If the mass of molecules becomes half and their velocities become double the pressu...The root mean square (rms) speed of oxygen molecules O2 at a certain temperature T (absolute) is υ. If the temperat...