KTGHard
Question
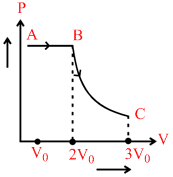
One mole of a gas is subjected to two process AB and BC, one after the other as shown in the figure. BC is represented by PVn = constant. We can conclude that (where T = temperature, W = work done by gas, V = volume and U = internal energy ).

Options
A.TA = TB = TC
B.VA < VB , PB < PC
C.WAB < WBC
D.UA < UB
Solution
(D)
Process AB is isobaric [V α T]
TB > TA ∴ UB > UA
WBC < WAB (Area under P-V curve)
Process AB is isobaric [V α T]
TB > TA ∴ UB > UA
WBC < WAB (Area under P-V curve)
Create a free account to view solution
View Solution FreeMore KTG Questions
A diatomic gas molecules has translational, rotational and vibrational degrees of freedom, then ratio is...One mole of an ideal gas at a temperature T1K expands slowly according to the law = constant. Its final temperature is T...For an ideal gas, the heat capacity at constant pressure is larger than than that at constant volume because...Equal volumes of two monoatomic gases, A and B, at same temperature and pressure are mixed. The ratio of specific heats ...250 litre of an ideal gas is heated at constant pressure from 27o C such that its volume becomes 500 liters. The final t...