KTGHard
Question
In a cyclic process shown on the P − V diagram the magnitude of the work done is :
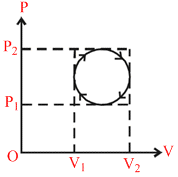

Options
A.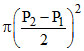
B.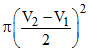
C.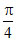 (P2 − P1) (V2 − V1)
(P2 − P1) (V2 − V1)
D.π (P2V2 − P1V1)
Solution
W.D. = π × Pressure Radius × volume Radius (area of ellipse)
W = π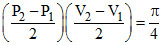
W = π
Create a free account to view solution
View Solution FreeMore KTG Questions
The pressure of an ideal gas is written as E = . Here E stands for...A cyclic process ABCD is shown in the P−V diagram. (BC and DA are isothermal)Which of the following curves represe...For the molecules of an Ideal gas, Which of the following velocity average can not be zero...If temperature is changed from 27oC to 327oC. The ratio of K.E. molecules at two temperature....Root mean square velocity for a certain di-atomic gas at room temperature 27oC is found to be 1930 m/sec. The gas is -...