KTGHard
Question
A cyclic process ABCD is shown in the P−V diagram. (BC and DA are isothermal)
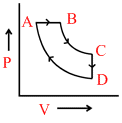
Which of the following curves represents the same process?

Which of the following curves represents the same process?
Options
A.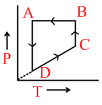

B.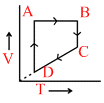

C.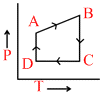

D.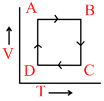

Solution
A → B constant pressure B → C T = constant
C → D constant Volume D → A T = constant
C → D constant Volume D → A T = constant
Create a free account to view solution
View Solution FreeMore KTG Questions
The ratio of the specific heats = γ in terms of degrees of freedom (n) is given by :...In figure, P-V curve of an ideal gas is given. During the process, the cumulative work done by the gas...The root mean square (rms) speed of oxygen molecules O2 at a certain temperature T (absolute) is υ. If the temperat...A diatomic ideal gas undergoes a thermodynamic change according to the P−V diagram shown in the figure. The total ...28 gm of N2 gas is contained in a flask at a pressure 10 atm. and at a temperature of 57oC. It is found that due to leak...