KTGHard
Question
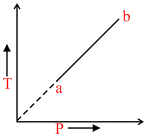
An ideal gas changes from state a to state b as shown in Fig. What is the work done by the gas in the process ?

Options
A.zero
B.positive
C.negative
D.infinite
Solution
T α P or P/T = constant
As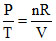 = constant or V = constant ∴ W = 0.
= constant or V = constant ∴ W = 0.
As
Create a free account to view solution
View Solution FreeMore KTG Questions
Average kinetic energy of a gas molecule depends on-...Supposing the distance between the atoms of a diatomic gas to be constant, its specific heat at constant volume per mole...Two containers A and B contain molecular gas at same temperature with mases of melecules are mA and mB, then relation of...Which of the following statement is not according to the postulates of kinetic theory of gases:-...Four curves A, B, C and D are drawn in the Fig. for a given amount of gas. The curves which represent adiabatic and isot...