Boron and Carbon FamilyHard
Question
If the unit cell of a mineral has cubic close packed (ccp) array of oxygen atoms with m fraction of octahedral holes occupied by aluminium ions and n fraction of tetrahedral holes occupied by magnesium
ions m and n respectively, are -
ions m and n respectively, are -
Options
A.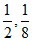
B.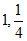
C.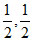
D.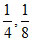
Solution
Effective number of O-2 = 4
Effective number of Al+3 = 4 m
Effective number of Mg+2 = 8 m
⇒ By charge balance 12 m + 16 n = 8
3 m + 4 n = 2
Possible value of m and n from given equation are
m =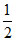 ; n =
; n = 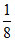
Effective number of Al+3 = 4 m
Effective number of Mg+2 = 8 m
⇒ By charge balance 12 m + 16 n = 8
3 m + 4 n = 2
Possible value of m and n from given equation are
m =
Create a free account to view solution
View Solution FreeMore Boron and Carbon Family Questions
Number of atoms in 558.5 gram Fe (at.wt. of Fe = 55.85 g mol-1) is...B(OH)3 + NaOH ⇋ NaBO2 + Na[B(OH)4] + H2O How can this reaction is made to proceed in forward direction?...The correct statement(s) for orthoboric acid is/are...Heating an aqueous solution of aluminium chloride to dryness will give...Which of the following reaction(s) will have final product as hydrocarbon -...