JEE Advanced | 2014Boron and Carbon FamilyHard
Question
The correct statement(s) for orthoboric acid is/are
Options
A.It behaves as a weak acid in water due to self ionization
B.Acidity of its aqueous solution increases upon addition of ethylene glycol
C.It has a three dimensional structure due to hydrogen bonding
D.It is a weak electrolyte in water
Solution
H3BO3 + H2O ⇋ B(OH)4+ + H+
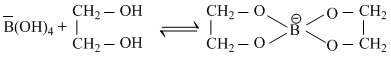
In boric acid, planar H3BO3 units are joined by hydrogen bonds to give layered structure.
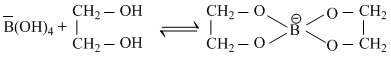

In boric acid, planar H3BO3 units are joined by hydrogen bonds to give layered structure.
Create a free account to view solution
View Solution FreeMore Boron and Carbon Family Questions
Which is non linear :-...From the following, the least stable structure is :...In the isolation of metals, reaction process usually results in :...Name the type of the structure of silicate in which one oxygen atom of [ SiO3]4- is shared ?...For making good quality mirrors, plates of float glass are used. These are obtained by floating molten glass over a liqu...