Chemical BondingHard
Question
Which of the following statements is/are correct ?
Commercial HCl is prepared by heating NaCl with H2SO4.
2NaCl + H2SO4 → N2SO4 + 2HCl
Commercial HCl is prepared by heating NaCl with H2SO4.
2NaCl + H2SO4 → N2SO4 + 2HCl
Options
A.196.0 g of pure H2SO4 is required for thr production of 245.0 g of cone HCl containing 40% HCl by weight
B.245.0 g of 80% H2SO4 by weight is required for the production of 365.0 g of cone. HCl containing 40% HCl by weight.
C.2 mol of pure H2SO4 is required for the production of 365 g of 40% HCl.
D.2.5 mol of 80% H2SO4 is required for the production of 365.0 g of 40% HCl.
Solution
a. Weight of 40% HCl = 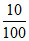 × 36.5 = 146 g
× 36.5 = 146 g
2 mol HCl (2 × 36.5 = 73 g) = 1 mol H2SO4 (98g)
Weight of H2SO4 = (146 g HCl)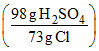
= 196 g H2SO4.
b. Weight of 80% H2SO4
=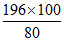 = 245 g H2SO4
= 245 g H2SO4
c. Mole of pure H2SO4
=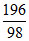 = 2 mol H2SO4
= 2 mol H2SO4
d. Mole of 80% H2SO4 =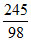 = 2.5 mol H2SO4
= 2.5 mol H2SO4
2 mol HCl (2 × 36.5 = 73 g) = 1 mol H2SO4 (98g)
Weight of H2SO4 = (146 g HCl)
= 196 g H2SO4.
b. Weight of 80% H2SO4
=
c. Mole of pure H2SO4
=
d. Mole of 80% H2SO4 =
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following options represents the correct bond order ?...Which of the following does not contain any coordinate bond ?...At what temperature, the most probable speed of H2 will be same as that of root mean square velocity ofN2 at 300 K?...Match the catalysts to the correct processes :-...Which one of the following molecular hydrides acts as a Lewis acid ?...