Atomic StructureHard
Question
At temperatuere T, the average kinetic energy of any particle is 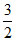 KT. The de Broglie wavelength follows the order :
KT. The de Broglie wavelength follows the order :
Options
A.Visible photon > Thermal neutron > Thermal electron
B.Thermal proton > Thermal electon > Visible photon
C.Thermal proton > Visible photon > Thermal electron
D.Visible photon > Thermal electron > Thermal neutron
Solution
De-broglie wavelength (for particles) = 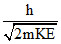
As temperature is same, KE is same. So, λ ∝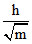
Hence λdb (electron) > λdb (neutron)
As temperature is same, KE is same. So, λ ∝
Hence λdb (electron) > λdb (neutron)
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The uncertainty in measuring the speed of a particle is zero. Uncertainty in measuring its position will be...In H-atom, if ′x′ is the radius of the first Bohr orbit, de Broglie wavelength of an electron in 3rd orbit i...An isotone of Ge is...The orbital angular momentum of a 4p electron will be...Light of wavelength (λ) strikes a metal surface with intensity X and the metal emits Y electrons per second of maximum k...