Atomic StructureHard
Question
In H-atom, if ′x′ is the radius of the first Bohr orbit, de Broglie wavelength of an electron in 3rd orbit is :
Options
A.3 π x
B.6 π x
C.9x/2
D.x/2
Solution
r1 = 0.529 (Ao)
r3 = 0.529 × (3)2 (Ao) = 9x
so, λ =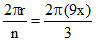 = 6 πx.
= 6 πx.
r3 = 0.529 × (3)2 (Ao) = 9x
so, λ =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The total spin resulting from a d7 configuration is :...If two py - orbital from two atoms are forming π - bond, then predict their approach axis-...M-2 configuration is 1s2 2s2 2p6 then M+2 configuration will be :-...Which of the following element is represented by electronic configuration1s22s22px12py12pz1 ?...Incorrect statement is :-...