AIPMT | 2015Atomic StructureHard
Question
The angular momentum of electron in ′d′ orbital is equal to :-
Options
A.√2 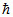
B.2√3 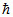
C.0 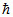
D.√6 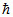
Solution
-Orbital angular momentum = 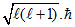
for d-orbital l = 2
so orbital angular momentum =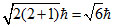
for d-orbital l = 2
so orbital angular momentum =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Photon of which light has maximum energy :...The correct set of four quantum numbers for the valence electrons of rubidium atom (Z = 37) is...Which of the following statement (s) are wrong ?...Rutherford′s experiment on scattering of α- particles showed for the first time that the atom has...For a d-electron, the orbital angular momentum is :...