Atomic StructureHard
Question
Which of the following statement (s) are wrong ?
Options
A.Photons having energy 400 kJ will break 4 mole bonds of a molecule A2 where A - A bond dissociation energy is 100kJ/mol.
B.Two bulbs are emitting light having wavelength 2000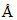 3000
3000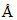 respectively. If the bulbs A & B are 40 watt and 30watt respectively then the ratio of no. of photons emitted by A & B per day is 1/2.
respectively. If the bulbs A & B are 40 watt and 30watt respectively then the ratio of no. of photons emitted by A & B per day is 1/2.
C.When an electron make transition from lower to higher orbit, photon is emitted.
D.None of the above
Solution
(A) Since the number of photons is not specified (it may or may not be equal to 4 NA). So, this statement is not always true.
(B) No.of photon emitted per day × Energy of one photon = Energy emitted per day.
For bulb A,neA ×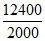 × 1.6 × 10-19 = 40 × 24 × 3600.
× 1.6 × 10-19 = 40 × 24 × 3600.
For bulb B, neB ×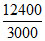 × 1.6 × 10-19 = 30 × 24 × 3600.
× 1.6 × 10-19 = 30 × 24 × 3600.
∴ neA : neB = 8 : 9.
(B) No.of photon emitted per day × Energy of one photon = Energy emitted per day.
For bulb A,neA ×
For bulb B, neB ×
∴ neA : neB = 8 : 9.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
According to Bohr′s model of hydrogen atom the electric current generated due to motion of electron in nth orbit i...Any p-orbital can accommodate upto :...When photons of energy 4.25 eV strikes the surface of a metal ‘A’, the ejected photoelectrons have maximum kinetic energ...For principle quantum number n = 4, the total number of orbitals having l = 3 is :...The value of Planck′s constant is 6.63 × 10-34 J-s.The speed of light is 3 × 1017 nm s-1. Which value is closest t...