AIPMT | 2015d and f-Block ElementsHard
Question
Magnetic moment 2.84 B.M. is given by :-
(At. nos, Ni = 28, Ti = 22, Cr = 24, Co = 27)
(At. nos, Ni = 28, Ti = 22, Cr = 24, Co = 27)
Options
A.Ti3+
B.Cr2+
C.Co2+
D.Ni2+
Solution
Ni+2 = 3d8 
number of unpaired electrons (n) =2
∵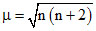
hence m = 2.8 B.M., paramagnetic

number of unpaired electrons (n) =2
∵
hence m = 2.8 B.M., paramagnetic
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
Of the following outer electronic configurations of atoms, the highest oxidation state is achieved by which one of them?...For which one of the following ions, the colour is not due to d-d transition :-...The existence of two different coloure complexes with the composition of [Co(NH3)4Cl2]+ is due to...The pair(s) of reagents that yield paramagnetic species is/are...In the manufacture of iron from an iron oxide ore, limestone is added because it acts as :-...