d and f-Block ElementsHard
Question
The existence of two different coloure complexes with the composition of [Co(NH3)4Cl2]+ is due to
Options
A.linkage isomerism
B.geometrical isomerism
C.coordination isomerism
D.ionisation isomerism
Solution
Complexes of [MA4B2] type exhibit geometrical isomerism.
The complex [Co(NH3)4Cl2]+ is a [MA4B2] type complex and thus, fulfils the conditions that are necessary to exhibit geometrical isomerism. Hence, it has two geometrical isomers of different colours as :
The structure of the geometrical isomers is as
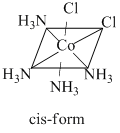
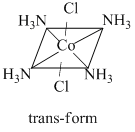
For linkage isomerism, presence of ambidentate ligand is necessary. For coordination isomerism, both the cation and anion of the complex must be complex ions. For ionisation isomerism, an anion different to the ligands must be present outside the coordination sphere. All these conditions are not satisfied by this complex. Hence, it does not exhibit other given isomerisms.
The complex [Co(NH3)4Cl2]+ is a [MA4B2] type complex and thus, fulfils the conditions that are necessary to exhibit geometrical isomerism. Hence, it has two geometrical isomers of different colours as :
The structure of the geometrical isomers is as


For linkage isomerism, presence of ambidentate ligand is necessary. For coordination isomerism, both the cation and anion of the complex must be complex ions. For ionisation isomerism, an anion different to the ligands must be present outside the coordination sphere. All these conditions are not satisfied by this complex. Hence, it does not exhibit other given isomerisms.
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
The atomic numbers of vanadium (V), Chromium (Cr), manganese (Mn) and iron (Fe) are respectively 23, 24, 25 and 26. Whic...Lanthanoid contraction is caused due to...[Fe(H2O)6]+2 and [Fe(CN)6]4- differ in :-...A pink coloured salt turns blue on heating. The presence of which cation is most likely ?...Which one is paramagnetic :-...