AIPMT | 2015Atomic StructureHard
Question
The species Ar, K+ and Ca2+ contain the same number of electrons. In which order do their radii increase ?
Options
A.Ca2+ < Ar < K+
B.Ca2+ < K+ < Ar
C.K+ < Ar < Ca2+
D.Ar < K+ < Ca2+
Solution
In isoelectronic species
Atomic radius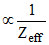
hence increasing order of radius is Ca+2 < K+ < Ar
Atomic radius
hence increasing order of radius is Ca+2 < K+ < Ar
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Energy of an electron is given by E = - 2.178 × 10-18 J Wavelength of light required to excite an electron in an hy...For the following compounds, the correct statement(s) with respect to nucleophilic substitution reactions is(are)...What are the number of atoms per unit cell and the number of nearest neighbours in a body centered cubic structure?...The number of unpaired electron in G. S., first E.S. and second E.S. of S (Z = 16) are, respectively,...Last line of Lyman series for H-atom has wavelength λ1, 2nd line of Balmer series has wavelength λ2 Å then...