Atomic StructureHard
Question
Last line of Lyman series for H-atom has wavelength λ1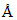 , 2nd line of Balmer series has wavelength λ2 Å then
, 2nd line of Balmer series has wavelength λ2 Å then
Options
A.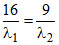
B.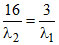
C.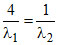
D.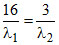
Solution
∴ λ1 =
∴
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
How many of the following compounds have used their d-orbital in hybridisationCF4, SF4, I3-, SOF4, PF3Cl2, SnCl2, SO42-...Photon having wavelength 310 nm is used to break the bond of A2 molecule having bond energy 288 kJ mol-1 then % of energ...The value of Rydberg constant R, if He+ ions are known to have the wavelength difference between the first (of the longe...To what series does the spectral line of atomic hydrogen belong if its wave number is equal to the difference between t...For which of the following element, all of its existing ion Mx+ will be diamagnetic?...