JEE Advanced | 2013Coordination CompoundHard
Question
Consider the following complex ions P, Q and R ,
P = [FeF6]3- , Q = [V(H2O)6]2+ and R = [Fe(H2O)6]2+
The correct order of the complex ions, according to their spin-only magnetic moment values (in B.M.)is -
P = [FeF6]3- , Q = [V(H2O)6]2+ and R = [Fe(H2O)6]2+
The correct order of the complex ions, according to their spin-only magnetic moment values (in B.M.)is -
Options
A.R < Q < P
B.Q < R < P
C.R < P < Q
D.Q < P < R
Solution
spin only magnetic moment 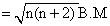
n → number of unpaired electrons
P : [FeF6]3-
F- → (weak field ligand)
ᐃ0 < P
Fe3+ ; [Ar] 3d5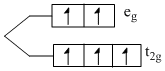
number of unpaired electron = 5
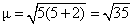
Q : [V(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
V2+ → [Ar]3d3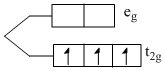
number of unpaired electron = 3
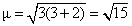
R : [Fe(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
Fe2+ ; [Ar] 3d6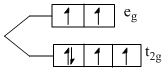
number of unpaired electron = 4
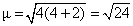
Hence, Ans (B) : Q < R < P
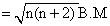
n → number of unpaired electrons
P : [FeF6]3-
F- → (weak field ligand)
ᐃ0 < P
Fe3+ ; [Ar] 3d5

number of unpaired electron = 5
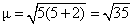
Q : [V(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
V2+ → [Ar]3d3

number of unpaired electron = 3
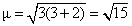
R : [Fe(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
Fe2+ ; [Ar] 3d6

number of unpaired electron = 4
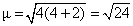
Hence, Ans (B) : Q < R < P
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
How many isomers are possible for the complex ion [Cr(NH3)Cl3(OH)2]2- ?...The total number of possible isomers of the compound [CuII(NH3)4] [PtIICl4] are:...Geometrical shapes of the complexes formed by the reaction of Ni2+ with Cl-, CN- and H2O, respectively, are...Malachite is an ore of :...Which of the following statements is correct with respect to the metal carbonyls of Ist transition series?...