Chemical BondingHard
Question
Which one of the following molecules is expected to exhibit diamagnetic behaviour ?
Options
A.C2
B.N2
C.O2
D.S2
Solution
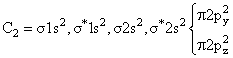 (Diamagnetic)
(Diamagnetic) 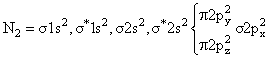 (Diamagnetic)
(Diamagnetic)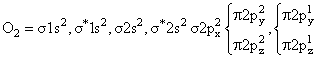 (Paramagnatic)
(Paramagnatic)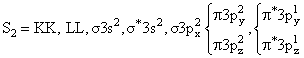 (Paramagnetic)
(Paramagnetic)C2 & N2 both are diamegnatic actully, but according to the language of question,expected to be diamegnatic.
If we consider energy leval diagram of C2 and N2 similar to O2 then C2 become paramagnetic & N2 remain diamegnatic so best answer of Question is N2.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Ease of oxidation in following compounds will be - (I) (II) (III)...In which pair of species the hybrid state of B is sp3 :-...The correct order of bond angles (smallest first) in H2S, NH3, BF3 and SiH4 is...The molecules that will have dipole moment are...In which of the following pairs the two species are not isostructural ?...