SolutionHard
Question
A solution of Na2S2O3 is standardized iodometrically against 0.167 g of KBrO3 where BrO3- changesto Br-. This process requires 45 mL of the Na2S2O3 solution. What is the strength of the Na2S2O3 ?
[Mw. of KBrO3 = 167]
[Mw. of KBrO3 = 167]
Options
A.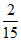 N
N
B.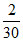 N
N
C.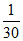 N
N
D.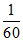 N
N
Solution
Eq. of Na2S2O3 ⇒ Eq. of KBrO3
N × 45 =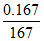 × 6 × 1000
× 6 × 1000
N =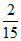
M =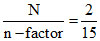
N × 45 =
N =
M =
Create a free account to view solution
View Solution FreeMore Solution Questions
Consider the following four liquids :1. Water : highly polar ; H-bonding2. Hexanol : slightly polar ; some H-bonding3. C...The ratio between lowering of vapour pressure of solution and mole fraction of solute is equal to...Water and chlorobenzene are immiscible liquids. Their mixture boils at 90°C under a reduced pressure of 9.031 × 104 Pa. ...Which of the following liquid pairs do not show positive deviation from Raoult’s law?...Observe the P-T phase diagram for a given substance A. Then melting point of A(s), boiling point of A(l), critical point...