ThermochemistryHard
Question
50.0 mL of 0.10 M HCl is mixed with 50.0 mL of 0.10 M NaOH. The solution′s temperature rises by 3.0oC. Calculate the enthalpy of neutralization per mole of HCl.
Options
A.-2.5 × 102 kJ/mole
B.-1.3 × 102 kJ/mole
C.-8.4 × 101 kJ/mole
D.-6.3 × 101 kJ/mole
Solution
Assuming density of solution is 1g/cc
and specipic heat is 4.2 J/gc
q = msᐃT = 100 × 4.2 × 3
millimoles of acid nuetralized = 5
ᐃH = - 100 × 4.2 × 3 ×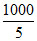 = - 2.52 × 102 kJ/mole.
= - 2.52 × 102 kJ/mole.
and specipic heat is 4.2 J/gc
q = msᐃT = 100 × 4.2 × 3
millimoles of acid nuetralized = 5
ᐃH = - 100 × 4.2 × 3 ×
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
AB, A2 and B2 are diatomic molecules. If the bond enthalpies of A2, AB and B2 are in the ratio 2:2:1 and enthalpy of for...Based on the following thermochemical equations,H2O(g) + C(s) → CO(g) + H2(g): ΔH = 131 kJCO(g) + ½ O2(g) → CO2(g): ΔH =...Benzene burns in oxygen according to the following reactions.C6H6(l) + 15/2 O2(g) → 3H2O(l) + 6CO2(g)If the standard ent...The enthalpy of formation of methane(g) at constant pressure is −18,500 cal/mol at 27°C. The enthalpy of formation at co...One gram sample of NH4NO3 is decomposed in a bomb calorimer, the temperature of the calorimeter increase by 6.12 K. The ...