Ionic EquilibriumHard
Question
Which of the following is incorrect :-
Options
A.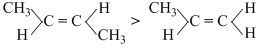
(Stability)
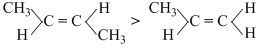
(Stability)
B.H-COOH > CH3-COOH
(Reactivity for esterification)
(Reactivity for esterification)
C.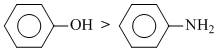
(Reactivity for coupling reaction in alkaline medium)
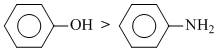
(Reactivity for coupling reaction in alkaline medium)
D.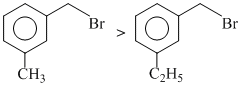
(Reactivity for hydrolysis)

(Reactivity for hydrolysis)
More Ionic Equilibrium Questions
The dissociation constant of acetic acid is 0.000018 and that for cyano acetic acid is 0.0036 at 298 K. What would be th...The solubility product Mg(OH)2 in water at 25oC is 2.56 × 10-13(mol/lt)3 while that of Al(OH)3 is 4.32 × 10-34...Small amount of freshly precipitated magnesium hydroxides are stirred vigorously in a buffer solution containing 0.25 M ...The degree of dissociation of water at 25°C is 1.8 × 10–7% and the density is 1.0 g cm–3. The ionic product for water is...To a solution of acetic acid, solid sodium acetate is gradually added. When ‘x g’ of the salt has been added, the pH has...