Ionic EquilibriumHard
Question
The solubility product Mg(OH)2 in water at 25oC is 2.56 × 10-13(mol/lt)3 while that of Al(OH)3 is 4.32 × 10-34 (mol/lt)4. If s1 and s2 are the solubilities of Mg(OH)2 and Al(OH)3 in water in mol/lt at 25oC, what is the ratio, s1/s2 ?
Options
A.2 × 105
B.2 × 104
C.3 × 106
D.3 × 103
Solution
Ksp of Mg (OH)2 = 2.56 × 10-13
4 s13 = 2.56 × 10-13 ⇒ s1 = 4 × 10-5 M
Ksp of Al (OH)3 = Ksp = 4.32 × 10-34
27 s24 = 4.32 × 10-34 ⇒ s2 = 2 × 10-9 M
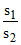 =
= 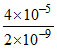 = 2 × 104
= 2 × 104
4 s13 = 2.56 × 10-13 ⇒ s1 = 4 × 10-5 M
Ksp of Al (OH)3 = Ksp = 4.32 × 10-34
27 s24 = 4.32 × 10-34 ⇒ s2 = 2 × 10-9 M
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A sample of AgCl was treated with 5.00 ml of 2.0 M Na2CO3 solution to give Ag2CO3. The remaining solution contained 0.00...Which of the following statements about a weak acid strong base titration is/are correct ?...Concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 ×10-4 mol L-1 Solubility product of Ag2C2O4...Ammonia forms the complex ion [Cu(NH3)4]2+ with copper ions in alkaline solutions but not in acidic solutions. What is t...The pH of an aqueous solution of 1.0 M ammonium formate assuming complete dissociation is (pKa of formic acid = 3.8 and ...