ElectrochemistryHard
Question
A hydrogen gas electrode is made by dipping platinum wire in a solution of HCl of pH = 10 and by passing hydrogen gas around the platinum wire at one atm pressure. The oxidation potential of electrode would be ?
Options
A.1.81 V
B.0.059 V
C.0.59 V
D.0.118 V
Solution
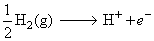
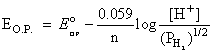
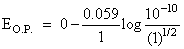
(pH = 10, [H+] = 10-10 M)
EO.P. = 0.59 V
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Three faradays of electricity was passed through an aqueous solution of iron (II) bromide. The mass of iron metal (at. m...How many faradays are required to reduce one mol of MnO4- to Mn2+ -...CH3O - CH = CH - NO2 ICH2 = CH - NO2 IICH2 = CH - Cl IIICH2 = CH2 IV Which of the following is the correct order of C - ...In the reaction I2 = I- → I3-, the Lewis base is :...Which of the following pair of metals, when coupled, will give maximum EMF for a voltaic cell?...