Gaseous StateHard
Question
What is the total pressure exerted by the mixture of 7.0 g of N2, 2g of hydrogen and 8.0 g of sulphur dioxide gases in a vessel of 6 L capacity that has been kept in a reservoir at 27oC?
Options
A.2.5 bar
B.4.5 bar
C.10 atm
D.5.7 bar
Solution
No. of moles of N2 = 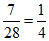
No. of moles of H2 = 1 Mole Total moles =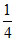 + 1 +
+ 1 + 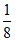
No. of moles of SO2 =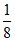 moles =
moles = 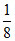 (2 + 8 + 1) =
(2 + 8 + 1) = 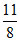
P =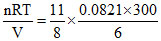 = 5.64 ≈ 5.7 atm.
= 5.64 ≈ 5.7 atm.
No. of moles of H2 = 1 Mole Total moles =
No. of moles of SO2 =
P =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Temperature at which r.m.s. speed of O2 is equal to that of neon at 300 K is :...Chemical temperature for A, B, C and D gases are 25oC, 10oC, -80oC and 15oC respectively. which gas will be liquefied mo...Which of the following is/are correct for an mono atomic ideal gas molecules :...If Z is a compressibility factor, van der Waals equation at low pressure can be written as...In the figure-2 the pressure of the confined gas will be...