Chemical Kinetics and Nuclear ChemistryHard
Question
The rate constant of a reaction is given by K = 2.1 ×1010 exp (-2700 / RT) it means that is :
Options
A.log K vs 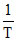 will be a straight line with slope =
will be a straight line with slope = 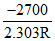
B.log K vs 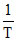 will be a straight line with intercept on log K axis = log 2.1 × 1010
will be a straight line with intercept on log K axis = log 2.1 × 1010
C.The number of effective collisions are 2.1 × 1010 cm-3s-1
D.(1) & (2) both
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Units of rate constant of first and zero order reactions in terms of molarity M unit are respectively...At 370 K a gaseous reaction X → 2Y + Z is found to be first order. Starting the reaction from pure X the total pre...The rate law for the reaction RCI + NaOH(aq) → ROH + NaCl is given by, rate = K1[RCl]. The rate of the reaction will be1...Select the correct statements out of I, II and III for zero order reactionI : Quantity of the product formed is directly...Assuming that the charged particles emitted during natural decay of 92U235 atoms are a and b particles only, which of th...