Basic Maths and Units and DimensionsHard
Question
An ideal gas is expanding such that PT2 = constant. The coefficient of volume expansion of the gas is:
Options
A.1/T
B.2/T
C.3/T
D.4/T
Solution
PT2 = constant and PV = nRT
implies V ∝ T3
⇒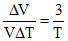
⇒ γ =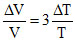
implies V ∝ T3
⇒
⇒ γ =
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
Two identical glass rods S1 and S2 (refractive index = 1.5) have one convex end of radius of curvature 10 cm. They are p...Two forces are such that the sum of their magnitudes is 18 N and their resultant is 12 N which is perpendicular to the s...In the figure ABC is the cross section of a right angled prism and BCDE is the cross section of a glass slab. The value ...A wind with speed 40 m/s blows parallel to the roof of a house. The area of the roof is 250 m2. Assuming that the pressu...An observer can see through a pin-hole the top end of a thin rod of height h, placed as shown in the figure. The beaker ...