Chemical BondingHard
Question
Which of the following has the minimum bound length ?
Options
A.O2-
B.O22-
C.O2
D.O2+
Solution
Bond order of O2+ = 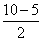 = 2.5
= 2.5
Bond order of O2- =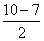 = 2.5
= 2.5
Bond order of O22- =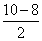 = 2
= 2
Bond order of O2 =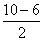 = 2
= 2
∵ Maximum bond order = minimum bond length.
∴ Bond length is minimum for O2+
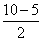 = 2.5
= 2.5 Bond order of O2- =
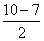 = 2.5
= 2.5Bond order of O22- =
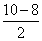 = 2
= 2Bond order of O2 =
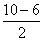 = 2
= 2∵ Maximum bond order = minimum bond length.
∴ Bond length is minimum for O2+
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Treatment of cyclopentanone with methyl lithium gives which of the following species?...-Match the following graphs :XA = 1 XB = 1 XB = 0 XA = 0GraphA (i) (+ deviation)B (ii) IdealC (iii) (-) deviation...The correct order of the stoichiometries of AgCl formed when AgNO3 in excess is treated with the complexs : CoCl3.6NH3, ...The molecule that has linear structure is :...In which of the following compounds, breaking of covalent bond take place :-...