SolutionHard
Question
One mole of a solute A is dissolved in a given volume of a solvent. The association of the solute take place as follows
nA ⇋ An
If α is the degree of association of A, the van′t Hoff factor i is expressed as :-
nA ⇋ An
If α is the degree of association of A, the van′t Hoff factor i is expressed as :-
Options
A.i = 1 - α
B.i = 1 + α/n
C. 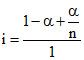
D.i = 1
More Solution Questions
When mercuric iodide is added to the aqeous solution of potassium iodide :...The limiting value of van’t Hoff factor for Na2SO4 is...The mass of KCl required to depress the freezing point of 500 g water by 2 K is (Kf = 1.86, K = 39)...The molecular weight of benzoic acid in benzene as determined by determined by depression in freezing point method corre...How many moles of sucrose should be dissolved in 500 gms of water so as to get a solution which has a difference of 104o...